SURFACE TREATMENT
Passivation process
Passivation process
What is passivation process?
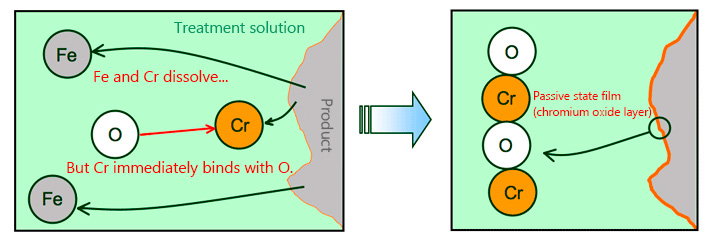
It refers to the chromium oxide layer which is formed on the surface as the chromium (Cr) contained in stainless steel binds with oxygen (O).
While iron (Fe) and chromium may be eluted as it is soaked in the treatment solution, chromium is extremely easy to oxidize and stable and thus quickly remains on surface as chromium oxide. Therefore, chromium concentration on surface increases in time, forming thicker chromium oxide layer. This is called the passive state film.Although it is called "film," it does not form a distinct film like plating.
Characteristics
- Processing is possible on stainless steel in general.
- It is colorless and transparent, causing no change in appearance after processing.
- There is also no change in dimensions. Therefore, it is possible to execute the process after providing designs on the base surface.
- Corrosion resistance is improved further by executing electrolytic polishing before passivation process.




